Results and Discussion

|
|
Figure 1
|

|
|
Figure 2
|

|
|
Figure 3
|
[6] The results of our analysis are presented in Figures 1, 2, and 3 for three seasons (winter, spring, and summer).
Shown are the mean diurnal profiles of hour average concentrations
of O3, SO2, NOx =NO+NO2, temperature,
relative humidity RH, short-wave irradiation UV-A, condensational sink CS,
and concentration of water vapor H2O for event
and nonevent days in Hyytiälä.
Winter
-
[7] In winter period the maximal levels of the trace gases concentrations were
[SO
2 ]=0.6 - 0.9ppb, [NOx ]= 1.5 - 3.5 ppb, RH=80 - 100%, [O3]=25 - 35 ppb,
at negative temperatures, minimal absolute humidity, and minimal levels of UV-A.
-
[8] A small amount of winter nucleation events is likely related to a deficiency of UV-A and O3
necessary for the formation of hydroxyl radicals, although the concentration of SO2 is normally very high.
-
[9] The particle formation processes are observed in increasing [O3] by 30%,
decreasing [NOx], increasing UV-A,
and at very small values of the condensation sinks (0.0004 1/s, see [Mäkela et al., 2000]).
-
[10] The diurnal dynamics of [SO2] regularly appears in the event days, with a minimum
in [SO2] at 15 h,
which well correlates with the maximums in UV-A, and T and a minimum in RH (80%).
-
[11] The particle formation process is often accompanied with the ozone consumption.
Spring
-
[12] The maximal frequency of event days is observed in spring time, with the growth rate being 2 - 5 nm/h.
-
[13] Maximal concentrations of O3 ([O3]=35 -47 ppb)
are typical for spring time as well as substantial enhancement in T, UV-A.
-
[14] The emission of volatile organic carbon grows due to the spring plant recovery and the beginning of the
photosynthetic process. The relation between [SO2] and VOC changes due to increasing in the bioactivity of trees.
-
[15] The formation of nucleation mode is accompanied with a decrease in [SO2] and [NOx]. The maximal
consumption of these gases are noticed at 14 - 16 h when a minimum in RH(43%) and a maximum in [O3] (47 ppb)
are observed.
-
[16] The average concentration levels of trace gases are much lower for event days, whereas the levels of O
3,
temperature, UV-A, are considerably higher than for nonevent days.
-
[17] A sharp difference is noticed between the daily variations to RH for the event days and those in nonevent days
(respective minimal RH are 40% and 80 - 90%) For nonevent days high relative humidity stimulates hygroscopic growth
of pre-existing aerosol particles which leads to their surface growth and thus the condensation sink which prevent the
formation of the nucleation mode. For event days RH decreases during the day from 75% down to 40%,
with the condensation sinks decreasing by the order-of-magnitude, from (4 - 5)
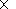 10-3s-1 to
(4 - 5)
10-3s-1 to
(4 - 5)
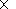 10-4s-1, whereas in
nonevent days the condensation sink varies within (2 -3)
10-4s-1, whereas in
nonevent days the condensation sink varies within (2 -3)
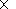 10-3s-1 - (2-3)
10-3s-1 - (2-3)
 10-2s-1 [Kulmala et al., 2004].
10-2s-1 [Kulmala et al., 2004].
Summer
-
[18] The number of event days is minimum, maximal particle growth rate reaches 5 - 10 nm/h.
-
[19] Very low concentrations of SO2 (0.2 - 0.1 ppb) NOx (0.7 - 0.3 ppb) are fixed in summer time,
whereas
the values of UV-A, T, and RH reach their maxima.
-
[20] High temperatures and UV-A substantially enhance the emission VOC, especially isoprene whose maximal
emission is observed in June and July. Because maximal emission of VOC coincides with minimal level of SO2 other
correlation links are expected to occur between different parameters.
-
[21] Nucleation events occur on dry days which agrees well to the data obtained by Buzorius et al., [2003],
where a negative correlation (-0.9) was found between the absolute water content and the particle formation
probability in Hyytiälä [Suni et al., 2003]. Likely this anticorrelation is a consequence of a sharp decrease of
the particle formation rate
due to the ozonolysis exocyclic monoterpenes in increasing the absolute water content [Bonn et al., 2002].
-
[22] Increasing the emission of VOC likely explains the maximal values of the particle growth rate up to
8 - 17 nm/h [Mäkela et al., 2000], maximal observed particle diameter (up to 60 nm), the latter being a growing function
of T.
-
[23] The highest concentrations of VOC and their further conversion to the non-volatile oxidized organic
precursors as well as growth of T and UV-A give rise to the particles become able to reach the Aitken or even
accumulation size, but the concentration of nucleation particles decreases with strong increase in VOC emission,
as supported by model experiments [Pirjola, 1999].

Powered by TeXWeb (Win32, v.2.0).




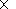 10-3s-1 to
(4 - 5)
10-3s-1 to
(4 - 5)
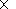 10-4s-1, whereas in
nonevent days the condensation sink varies within (2 -3)
10-4s-1, whereas in
nonevent days the condensation sink varies within (2 -3)
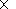 10-3s-1 - (2-3)
10-3s-1 - (2-3)
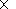 10-2s-1 [Kulmala et al., 2004].
10-2s-1 [Kulmala et al., 2004].
